Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
Infections are a serious health problem that affects millions of people each year. Among the infections, Urinary Tract Infection (UTI) is one of the major infections accounting for about 8.3 million visits to doctors yearly. Women are especially prone to UTIs. These are treated with antibacterial drugs. Emergence of pathogenic microorganisms that are resistant or multi resistant to a major class of antibiotics has increased in recent years due to indiscriminate use of synthetic antimicrobial drugs. In addition, high cost and adverse side effects are commonly associated with popular synthetic antibiotics (such as hypersensitivity, allergic reactions, etc.) and are serious burning global issues in treating infectious diseases (Jasmine etal., 2013). This has thus called for the search for alternative therapeutic options. Plants are the oldest source of pharmacologically active compounds and have provided humankind with various medically useful compounds for centuries (Mbwambo et al., 2007). Today it is estimated that more than two thirds of the world’s population relies on plant derived drugs. Among these plants, Moringa oleifera is a popular plant that has been reported to have an impressive range of medicinal uses with high nutritional value. Traditionally, M. oleifera have known for its coagulative properties on waste water, cardiac and circulatory stimulant, antitumor, antipyretic, antiepileptic, anti-inflammatory, antiulcer, antispasmodic, diuretic, antihypertensive, cholesterol lowering, antioxidant, antidiabetic, hepatoprotective, antibacterial, antifungal activities and have lots of macro and microelements and various forms of nutrients (USDA, 2003; Katayon et al., 2005; Kebreab et al., 2005; Farooq et al., 2007). Due to all these untapped advantages, this study is designed to investigate the in vitro susceptibility profile of bacteria associated with UTI to the various extracts of M. oleifera.
Materials and Methods
1 Sample collection
Following informed consent, Mid-stream urine (MSU) samples contained in sterile universal containers were collected from all study participants at the University of Uyo health center and conveyed to the laboratory within two hours.
2 Isolation of UTI associated Bacteria
Urine samples were inoculated on Nutrient agar medium, MacConkey agar medium and Cystine Lactose Electrolyte Deficient (CLED) agar medium. The inoculated plates were incubated at 37 ºC for 18 – 24 hour after which the colonies observed on the plates were subjected to morphological and biochemical characterization to identify the isolates.
3 Characterization and Identification of the Bacteria Isolates
The bacterial isolates were characterized based on their morphological, biochemical and fermentative characteristics as described by Chessbrough, (2006). The obtained characteristics were compared with those given by Barrow and Feltham (2003a); Barrow and Feltham, (2003b) for identification of the various genera.

We are supplier of moringa oil and moringa leaf powder from Indonesia – Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
4 Collection and Preparation of Plant Extracts
Healthy disease free Moringa oleifera leaves were collected from the Postgraduate farm
of Botany Department, University of Uyo. The collected plant species were identified and confirmed by the botanist then taken to the laboratory for extraction. In the laboratory, the obtained M. olieferaleaves were cleaned, shade-dried at room temperature and ground into a fine powder with the help of anelectrical grinder. Fifty grams (50 g) of the powder wastaken in soxhlet apparatus and 200 ml of organicsolvents viz acetone was added separately to run for 24 hours. Excess solvent was removed from the filtrate extraction using a rotary vacuum evaporator. After the complete evaporation, the extractions were stored separately at 4 0C in amber colored airtight bottles. Crude extract of the leaves was also obtained.
5 Preparation of Disc for antibacterial activities
Sterile filter papers were perforated and divided into two groups. The first group was treated with the acetone plant extracts of concentration 10µl while the second group treated with 30µl of the extract. This procedure was also carried out for the crude plant extract. The two set were allowed to absorb the extract for one hour. The prepared discs were then dried in controlled temperature to remove excess of moisture and used for antibacterial activity.
6 Plant extract susceptibility test
The Kirby-Bauer modified disc diffusion technique as recommended by National Committee for Clinical Laboratory Standards (NCCLS) was used to determine the susceptibility of the isolates to the plant extracts. A broth culture of peptone water (500 ml) was prepared and 5ml, dispensed into Petri dishes and autoclaved and allowed to cool. The test organisms were inoculated and incubated for 24 hours. Mueller Hinton agar was prepared and 15 ml dispensed into sterile Petri dishes and allowed to solidify. Using a sterile swab stick, each for the different isolates, the cultures were obtained by dipping. They were then spread on the media; differently for all the isolates. After 5minutes the antibiotic discs were placed on the surface of the inoculated media and incubated for 24 hours at 37 ºC. They were then observed for zones of inhibition, which were measured in millimeters and interpreted using the National Committee for Clinical Laboratory Standards (NCCLS) (Cheesbrough, 2006).
7 Determination of Minimum Inhibitory Concentration (MIC)
The Minimum Inhibitory Concentration of the crude seed extract of Moringa oleifera was determined using the method of Greenwood (1989) as described by Geidamet al. (2007). Serial dilution of the extract at the concentrations of 10, 30, 50, 70 and 100 mg/ml respectivelywere used to determine minimum inhibitory concentration and recorded as the least concentration of the extract that completely inhibited the growth of the organisms.
Results
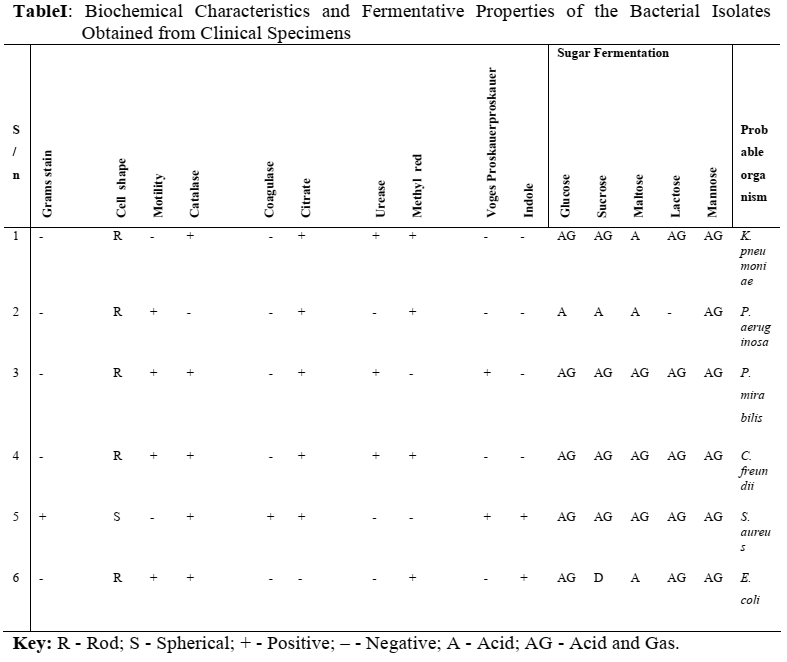
The morphological, biochemical and fermentative characteristics of the bacterial isolates obtained are reported on Table I. The isolates obtained from the clinical specimens consisted of one gram positive and five gram negative organisms. This table also identifies the most probable microorganisms based on the various characteristics obtained as described in Bergey’s Manual of Determinative Bacteriology to include Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus mirabilis, Citrobacter freundii, Staphylococcus aureus and Escherichia coli.
We are supplier of moringa oil and moringa leaf powder from Indonesia – Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
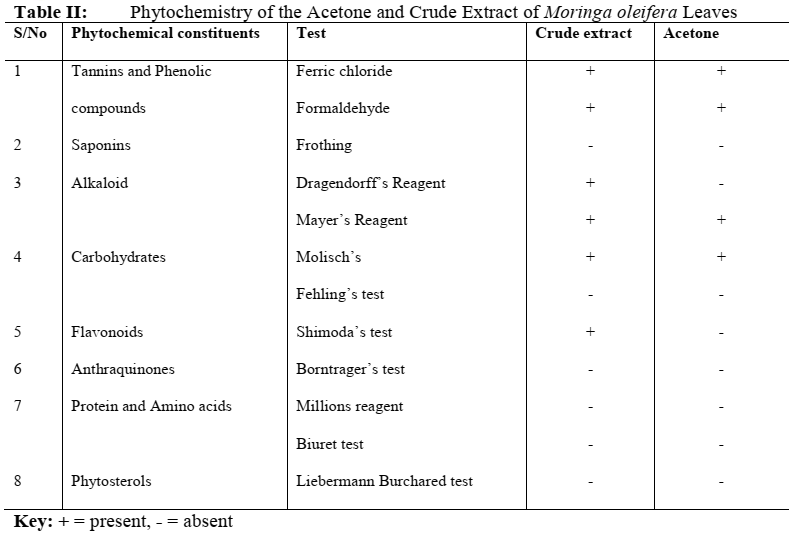
The Preliminary Phytochemical study carried out on the extract showed that the crude extract contained Flavonoids which was not found in the Acetone extract. Dragendorff’s reagent indicated the absence of alkaloid in the acetone extract but was found to be present when the acetone extract was screened using the Mayer’s reagent. Tannins and Phenolic compounds and carbohydrates were found to be present in the acetone and crude extract whereas Saponins, Anthraquinones, Protein and amino acids, and Phytosterols were absent in the extracts. A summary of the study is shown on Table II.
We are supplier of moringa oil and moringa leaf powder from Indonesia – Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
The result of the in vitro susceptibility profile of the bacterial isolates to 10mg/ml and 30mg/ml of the acetone and crude Moringa extract shows that, Pseudomonas aeruginosa wasmost susceptible to the extract with the diameter of halo zone of 12 mm and 15 mm; 10 mm and 13 mm for the 10 mg/ml and 30 mg/ml concentration of acetone and crude extract respectively. This was closely followed by Klebsiella pneumonia (8 mm and 12 mm for 10 mg/ml and 30 mg/ml of acetone and crude extract respectively) whereas Proteus mirabilis was found to be the most resistant among the isolates with no zone of inhibition and 2 mm at concentration of 10 mg/ml and 30 mg/ml of crude extract and diameter of 4mm and 6mm for acetone extract concentration of 10 mg/ml and 30 mg/ml respectively AS SHOWN ON Table III and IV.

We are supplier of moringa oil and moringa leaf powder from Indonesia – Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
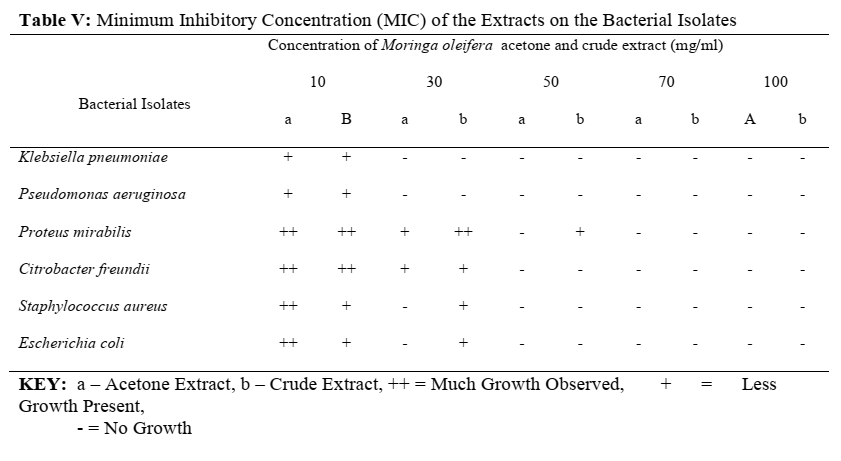
The results of the minimum inhibitory concentration (MIC) of the acetone and crude extracts on the bacterial isolates are also given below in Table V.
We are supplier of moringa oil and moringa leaf powder from Indonesia – Antibacterial effect of Moringa oleifera extracts on bacteria associated with urinary tract infection
Discussion
The preliminary phytochemical analysis of extract done also revealed that the plant provides a rich and rare combination of compounds which contribute to its therapeutic and high nutritional value. In comparison, the result of the phytochemical screening carried out on the extract showed great similarity to the result of the phytochemistry of the extract of the same plant carried out by Arun and Purnachandra (2011). However, this was centrally to finding of Auwalet al., (2013) who researched work on the Antibacterial and haematological activity of Moringa oleifera aqueous seed extract in Wistar albino rats as their aqueous seed extract of Moringa oleifera revealed high level of phytochemicals such as saponins, alkaloids, cardiac glycosides, flavonoids and anthraquinones. According to Jasmine et al., (2013), various parts of the plant such as the leaves, roots, seed, bark, fruit, flowers and immature pods act as cardiac and circulatory stimulants, possess antitumor, antipyretic, antiepileptic, anti-inflammatory, antiulcer, antispasmodic, diuretic, antihypertensive, cholesterol lowering, antioxidant, antidiabetic, hepatoprotective, antibacterial and antifungal activities (Jasmine et al., 2013).
The potency of the various extract increased as the concentration of the extract increased. This tends to correspond with the work of Arun and Purnachandra (2011), who in their work on; the phytochemical screening and antibacterial activity of Moringaoleifera lam. against Proteusmirabilis from urinary tract infected patients showed that the antibacterial activity of the extract on P. mirabilis increased as the concentration of the extract increased.
Apart from the increase in the antibacterial property of the extract with increase in concentration, it is also worth noting that the acetone extract had a greater antimicrobial activity than the crude extract. Among the isolates tested, P. aeruginosa was found to be most susceptible to the two concentrations (10mg/ml and 30 mg/ml) of the acetone and crude plant extracts used with diameter of clear zones of 12mm, 15mm and 10mm, 13mm respectively. This was followed by Klebsiella pneumonia 8mm, 12mm and 8mm, 10mm respectively. Of all the isolates, Proteus mirabilis was found to be least susceptible to the plant extracts. According to NCCLS standard, Proteus was resistant to the 10mg/ml concentration of the plant extract and 30 mg/ml concentration of the crude extract had little or insignificant effect on its growth, the acetone extract also had least effect on the growth of the organism. This might be attributed the fact that the organism has a vast metabolic mechanism which enables it reduce the pH of its growth medium (Brooks et al., 2004). It was noted that the resistance of P. mirabilis to the extract was followed by C. freundii. The susceptibility of S. aureus and Escherichia coli was also found to be higher in the acetone extract than the crude extracts. This might be attributed to the variation in the phytochemical property of the various extracts.
Conclusion
Based on the results obtained from this study and other published materials, it can be seen that various bacterial strains are associated with UTI and these include Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus mirabilis, Citrobacter freundii, Staphylococcus aureus and Escherichia coli. Although it was observed that extracts of Moringa oleifera had great antimicrobial effect on the various isolates, acetone extract of M. oleifera had a greater antibacterial property than the crude extract and the higher the concentration of the extract, the higher its antibacterial property.
For your inquiry about moringa oil/moringa powder :
Contact us :
E-mail: [email protected]
Phone: +6221-2903 4428
Fax: +6221-2903 4429
Sales Mobile/Whatsapp: +62813 8063 7778
Moringa Indonesia Fangardana is moringa oil company moringa oil wholesale moringa oil bulk supplier moringa oil manufacturer moringa oil producer moringa oil bulk seller moringa oil supplier moringa leaf powder organic moringa leaf powder moringa leaf company moringa leaf manufacturer moringa leaf seller moringa leaf supplier moringa leaf producer moringa leaf indonesia moringa oil indonesia